In the current drug manufacturing landscape, Pharmaceutical spray drying serves as a vital unit operation for converting liquid formulations into stable, high-quality solid powders. Largely defined by its speed and scalability, it enables the production of particles to meet stringent medical standards, from small laboratory batches to large quantities.
For manufacturers, the use of advanced Spray Dryers provides a solid technological approach to particle engineering, essential for ensuring the consistency and therapeutic efficacy of modern medications. By utilizing these systems, the industry creates complex particles with specific characteristics tailored to the delivery requirements of different active pharmaceutical ingredients (APIs).
What is the spray drying of pharmaceuticals?
The core concept of spray drying in pharmaceutical industry applications involves the rapid transformation of an atomized liquid feed into dry particles by exposure to hot, temperature-controlled air. This transformation is not merely a method of water removal but a sophisticated process of particle formation where dissolved or suspended solids release water from the liquid phase and solidify.
One of the most important advantages of this technique is its ability to protect heat-sensitive materials. As the water evaporates from the surface of an atomized droplet, it creates an evaporative cooling effect that maintains the product at a lower temperature despite exposure to high-temperature gas.
This mechanism is crucial for particle engineering, as it enables manipulation of a droplet’s “drying history” to achieve desired morphologies. For instance, by controlling the rate of surface solidification, manufacturers can produce particles with unique surface characteristics, such as hollow structures or high surface-to-volume ratios, which are essential for rapid reconstitution and improved solubility in the human body.
What are the methods of drying in the pharmaceutical industry?
The pharmaceutical sector employs several dehydration technologies, each selected based on the thermal sensitivity of the product and the required final structure. Traditional methods like tray drying or drum drying are often used for less sensitive materials, but frequently suffer from long processing times and potential non-uniformity in quality. Freeze drying (lyophilization) is widely regarded as the gold standard for extremely fragile biologics such as therapeutic proteins or vaccines, because it removes moisture via sublimation at very low temperatures. However, freeze drying is a time-consuming and energy-inefficient batch process that often lacks the precise control over particle size offered by other methods.
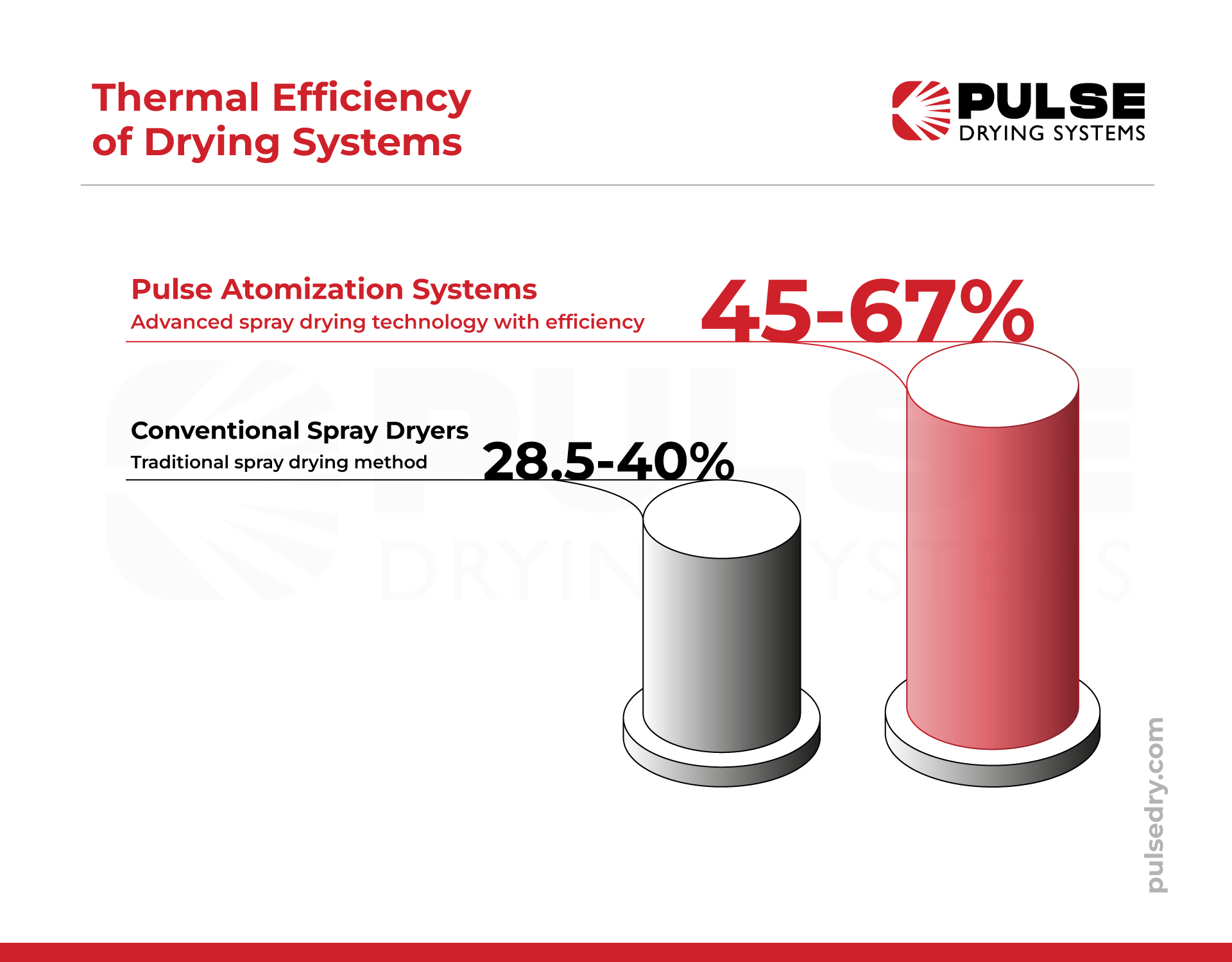
In contrast, pulse atomization spray drying has emerged as a high-efficiency continuous alternative. While conventional spray dryers typically operate at lower thermal efficiency ranges, advanced pulse systems can achieve significantly higher efficiency levels. This improvement is largely due to the use of high-velocity pulsating jets that intensify the rates of heat and mass transfer. These pulse systems allow for higher solids content in the feed without the need for dilution, which significantly lowers processing costs while maintaining high production rates.
What is the pharmaceutical spray drying process?
The technical pharmaceutical spray drying process comprises four primary stages: feed preparation/formulation, atomization, drying in hot air, and powder collection. The process begins with the liquid feed, which may contain APIs and essential excipients, being metered into the drying chamber. In advanced pulse atomization systems, “gas dynamic” atomization replaces traditional mechanical nozzles or rotary discs. High-velocity pulsating jets break the liquid into fine droplets with a tighter size distribution and more consistent morphology than traditional methods.
During the drying phase, these droplets encounter a controlled gas stream with inlet temperatures ranging from 200–425 °C. Despite these high temperatures, the residence time of the particles within the chamber is extremely short—typically less than five seconds—which prevents the product from reaching damaging temperature levels. To maintain GMP compliance and adhere to Quality by Design (QbD) standards, the outlet temperature is meticulously controlled within 70 and 105 °C to ensure a consistent drying environment and prevent hot spots that could lead to denaturation of the powder. Finally, the dried powder is separated from the gas using sanitary cyclones or baghouses, resulting in a free-flowing, stable product.
What is spray drying in pharma used for?
The application of spray drying in pharmaceutical industry settings is vast, addressing challenges in stability and drug delivery. A primary use is the improvement of drug bioavailability. By creating amorphous solid dispersions, the process can transform poorly soluble APIs into powders that dissolve much more readily, ensuring the patient receives the proper therapeutic dose.
Furthermore, the technology is indispensable for the production of biologics, including viral vaccines and heat-sensitive proteins. Because pulse atomization involves such rapid processing times, it allows for the successful dehydration of vaccines while retaining greater than 90% viability of the viral particles. This is a significant breakthrough for global health, enabling these vaccines to be stored and shipped in a stable dried state without the need for a continuous cold chain. The process is also utilized for encapsulation, shielding active ingredients from oxidation, and controlling their release profile to optimize therapeutic outcomes. By providing more homogeneous size distribution and superior particle characteristics, it ensures that every batch of medicine meets the highest safety and quality requirements.
Pulse Drying Systems is an upcoming innovator in providing advanced, GMP-compliant solutions for the pharmaceutical industry. Our proprietary pulse atomization technology is designed to reinvent the way that customers produce powders, offering unmatched results for APIs, biologics, and nutraceuticals. Contact us today to learn how our state-of-the-art systems can deliver the highest quality powders with significant performance advantages over traditional drying methods.
NOTE: The opening illustration appears to be AI-generated and is not accurate. Just use any photo of a pharma spray dryer instead. Thanks!







